History of the Environmental Protection Agency
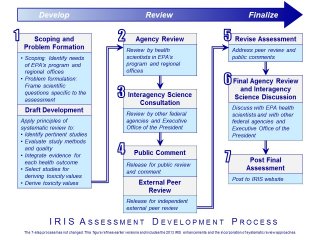 EPA’s mission is to protect human health and the environment. EPA’s IRIS Program supports this mission by identifying and characterizing the health hazards of chemicals found in the environment. Each IRIS assessment can cover a chemical, a group of related chemicals, or a complex mixture.
EPA’s mission is to protect human health and the environment. EPA’s IRIS Program supports this mission by identifying and characterizing the health hazards of chemicals found in the environment. Each IRIS assessment can cover a chemical, a group of related chemicals, or a complex mixture.
IRIS assessments:
- Are the preferred source of toxicity information used by EPA
- Are an important source of toxicity information used by state and local health agencies, other federal agencies, and international health organizations
The IRIS Program is located within EPA’s National Center for Environmental Assessment (NCEA) in the Office of Research and Development (ORD). The placement of the IRIS Program in ORD is intentional. It ensures that IRIS can develop impartial toxicity information independent of its use by EPA’s program and regional offices to set national standards and clean up hazardous sites.
IRIS Toxicity Values
IRIS assessments provide the following toxicity values for health effects resulting from chronic exposure to chemicals.
Oral is the amount of a chemical that one can ingest every day for a lifetime that is not anticipated to cause harmful noncancer health effects. The RfD can be compared to an estimate of exposure in mg/kg-day.
Inhalation is the concentration of a chemical that one can breathe every day for a lifetime that is not anticipated to cause harmful noncancer health effects. The RfC can be compared to an estimate of exposure concentration in mg/m3.
Cancer descriptors characterize the chemical as:
- Carcinogenic to Humans
- Likely to Be Carcinogenic to Humans
- Suggestive Evidence of Carcinogenic Potential
- Inadequate Information to Assess Carcinogenic Potential
- Not Likely to Be Carcinogenic to Humans
(OSF) is an estimate of the increased cancer risk from oral exposure to a dose of 1 mg/kg-day for a lifetime. The OSF can be multiplied by an estimate of lifetime exposure (in mg/kg-day) to estimate the lifetime cancer risk.
Inhalation (IUR) is an estimate of the increased cancer risk from inhalation exposure to a concentration of 1 µg/m3 for a lifetime. The IUR can be multiplied by an estimate of lifetime exposure (in µg/m3) to estimate the lifetime cancer risk.
What's the Role of IRIS Assessments in Risk Assessment?
Risk assessment is a four-step process described by the National Research Council (NRC) in 1983 as "the characterization of the potential adverse health effects of human exposures to environmental hazards." Characterizing risk involves integrating information on hazard, dose-response, and exposure.
An IRIS assessment includes the first two steps of the risk assessment process:
- Hazard Identification, which identifies credible health hazards associated with exposure to a chemical, and
- Dose-Response Assessment, which characterizes the quantitative relationship between chemical exposure and each credible health hazard. These quantitative relationships are then used to derive toxicity values.